
In regenerative medicine, mesenchymal stem cells (MSCs) act as “mobile repair factories.” They can differentiate into bone, cartilage, and fat cells, while also regulating immune responses and supporting tissue repair. Among them, human umbilical cord–derived MSCs (hUC-MSCs) show strong clinical promise thanks to their accessibility and low immunogenicity. However, these cells are highly sensitive to shear stress, prone to aggregation, and must maintain stemness during culture. Suboptimal conditions can stall growth, trigger premature differentiation, or even cause cell death.
The key to large-scale in-vitro expansion is creating a culture environment that is both gentle and highly efficient.
1.Core Characteristics of Mesenchymal Stem Cells
01 High Potential, Limited Quantity
MSCs occur naturally in very small quantities. A single clinical dose typically requires 10⁷–10¹⁰ cells, making large-scale in-vitro expansion essential while preserving their multipotent differentiation capacity.
02 Extreme Sensitivity to Shear Stress
As animal cells without protective cell walls, MSCs are highly vulnerable to fluid shear forces. Excessive shear stress can cause direct cell death, while even moderate levels may inhibit proliferation, induce premature differentiation, and reduce clinical effectiveness.
03 Strong Adhesion and Tendency to Aggregate
MSCs readily attach to microcarriers for growth and easily form cell clusters. When aggregate diameters exceed 1 mm, cells in the core can die from oxygen and nutrient deprivation. Large aggregates also make harvesting more difficult and reduce overall cell viability.
04 Stemness Must Be Strictly Maintained
Stemness—the cells’ multipotent differentiation potential—is their core value. Imbalances in shear force, aggregation, or nutrient supply during culture can suppress stemness gene expression and trigger premature differentiation into somatic cells.
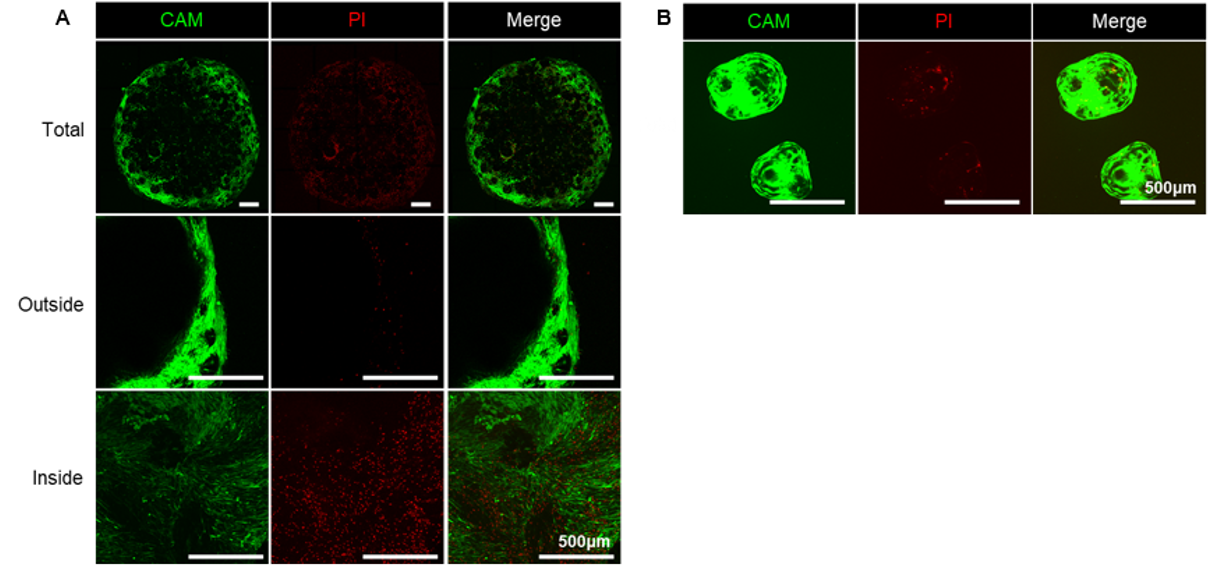
Figure 1. Mesenchymal Stem Cells Under the Microscope A. Oversized aggregates formed by excessive clustering, with extensive cell death in the center (red dots indicate dead cells); B. Healthy stem cells attached to individual microcarriers, showing uniform growth and optimal condition
2.Challenges of traditional bioreactors: Why Traditional Bioreactors Struggle to Effectively Culture Stem Cells?
Conventional spinner flasks rely on paddle stirring to keep cells suspended, but their flow-field design has inherent limitations. They cannot simultaneously control aggregation and minimize shear stress.
01 Low Rotation Speed Leads to Severe Aggregation
Lowering rotation speed (e.g., 30 rpm) to reduce shear stress allows microcarriers to settle, leading to rapid formation of large, millimeter-scale aggregates. After nine days of culture, average aggregate diameter can reach 3.36 ± 0.94 mm (see Figures 2A and 2B). These oversized clusters restrict oxygen and nutrient diffusion, causing internal cell hypoxia, nutrient depletion, and ultimately cell death.
02 High Rotation Speed Damages Stemness
Increasing rotation speed (e.g., above 60 rpm) helps reduce aggregate size to about 0.5 mm, but the resulting rise in shear stress suppresses cell proliferation and lowers final cell density. More importantly, elevated shear forces downregulate stemness genes and upregulate differentiation genes, weakening the cells’ multipotent differentiation capacity See Figures 2B and 2C.
03 Impeller Design Creates Chaotic Flow Fields
CFD simulations show that conventional flat-blade impellers generate multiple isolated vortices, fragmenting the flow field and causing uneven mixing. This chaotic environment can trap cells and microcarriers in vortices, worsening aggregation, or expose them to sustained high-shear forces. Adjusting rotation speed alone cannot deliver the gentle, uniform conditions required for stem cell culture See Figure 2D.
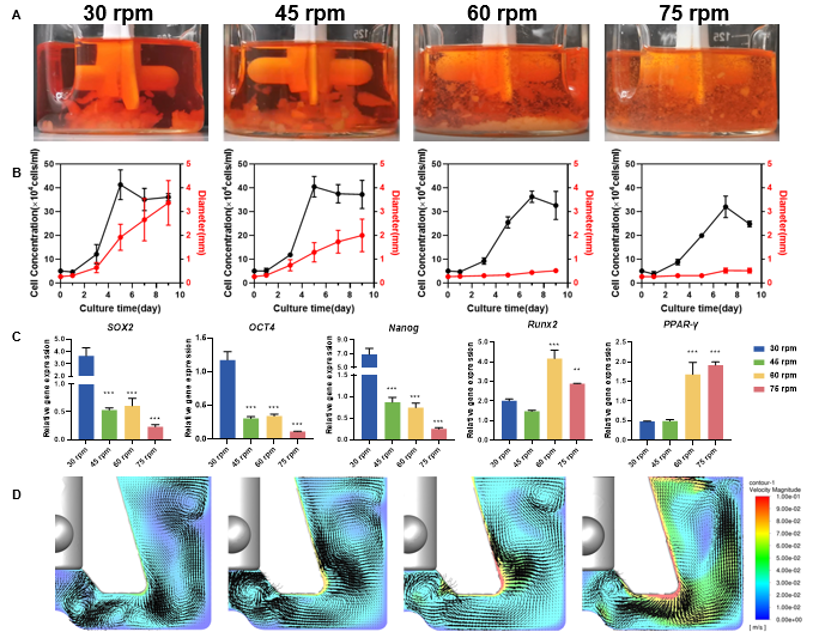
Figure 2. Key Limitations of Conventional Impellers A. Aggregate status on Day 9 at different rotation speeds: low speed leads to sedimentation and aggregation; high speed maintains suspension but introduces excessive shear stress; B. Effects of rotation speed on cell density and aggregate size: neither high nor low speed provides an optimal outcome; C. High speed downregulates stemness genes and upregulates differentiation genes; D. Flow fields generated by conventional impellers form multiple isolated vortices, resulting in poor mixing efficiency
3.Customized Bioreactor Enables High-Quality Expansion
Meeting the growth requirements of mesenchymal stem cells requires fundamental reactor design optimization. Using CFD modeling and structural redesign, a Modified Impeller (MI) was developed to overcome the limitations of conventional systems—improving flow-field dynamics, suspension performance, shear control, and overall culture outcomes.
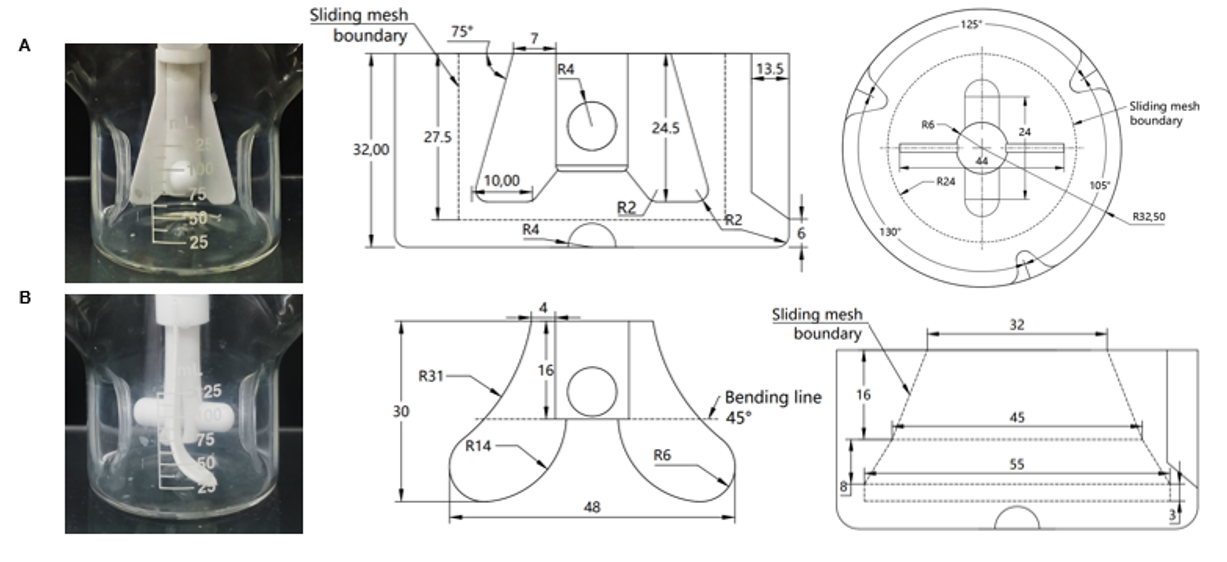
Figure 3. Schematic Comparison of the Conventional Impeller (A) and the Modified Impeller (B)
01 Optimization 1: Impeller Redesign for a Uniform, Low-Shear Axial Flow Field
Replacing the traditional radial-flow impeller with an axial-flow design (Figure 4A) creates full-vessel circulation, with culture medium moving top-to-bottom and bottom-to-top in a continuous loop. This conveyor-belt–like flow eliminates vortex dead zones, keeps cells and microcarriers evenly suspended, and significantly reduces localized shear stress.
02 Optimization 2: Quantitative Design to Balance Suspension and Shear Protection
At comparable average shear rates (SSRave), the new impeller increases axial pumping capacity (Qzave) by 50–60% versus conventional designs. This means substantially stronger suspension under equally gentle conditions. In practice, 45 rpm delivers the same suspension performance as 75 rpm with a traditional impeller—achieving true “low shear, high suspension” operation. See Figures 4B and 4C.
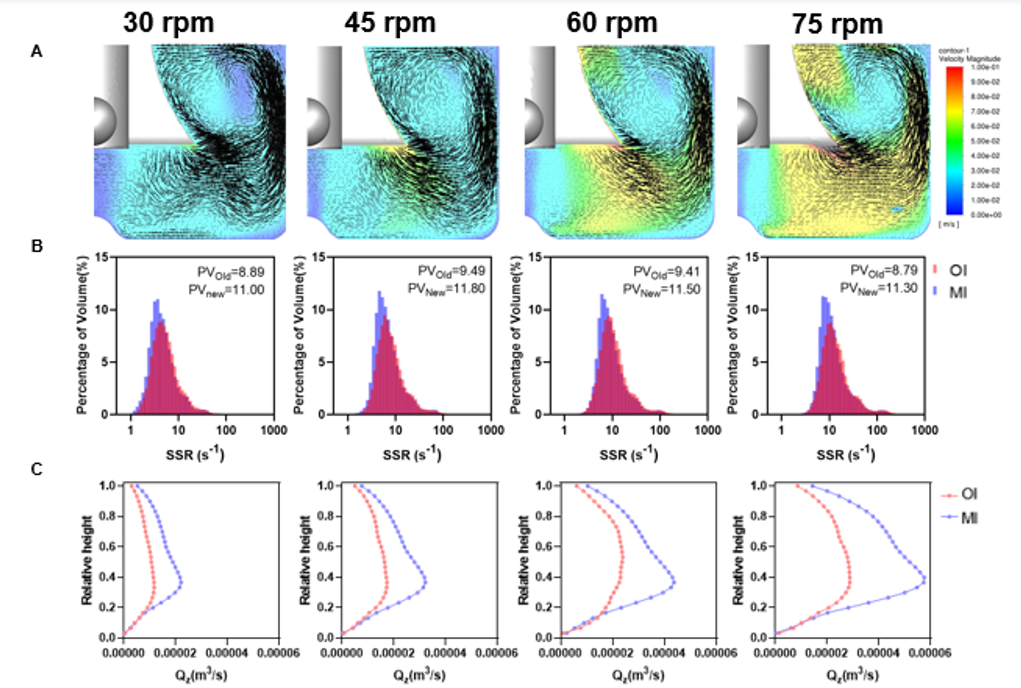
Figure 4. A. Fluid flow pattern in a spinner flask equipped with the improved impeller; B. Comparison of shear-rate volume fractions between conventional and new impellers across different zones; C. Qz distribution comparison between conventional and new impellers
03 Optimization 3: High-Quality, High-Density Expansion
When culturing mesenchymal stem cells with the new impeller:
Significantly reduced aggregation: Aggregate diameters are controlled within 1.25 mm at 30 rpm and 0.87 mm at 45 rpm, effectively preventing hypoxia-related cell death in aggregate cores.
Substantially higher cell density: Peak cell density increases by nearly 40%, reaching 5.771 × 10⁵ cells/mL at 30 rpm.
Stable stemness maintenance: Key stemness genes (e.g., SOX2, OCT4) are upregulated while differentiation genes are suppressed, preserving the cells’ regenerative potential.
Figure 5 clearly shows the improved culture performance enabled by the new impeller design. Cell–microcarrier aggregates are smaller and more uniformly distributed, proliferation curves show markedly improved growth, and pluripotency gene expression is better preserved.
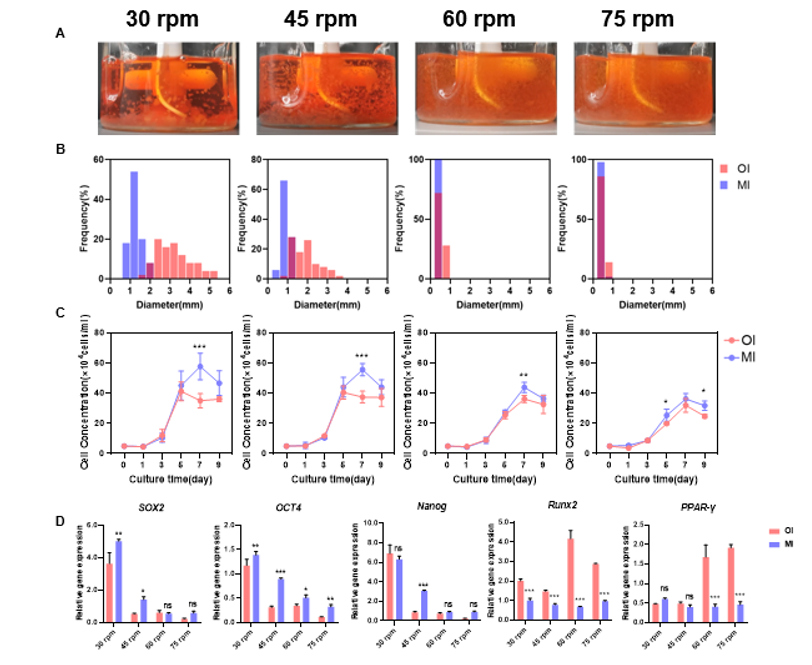
Figure 5. Performance Evaluation of the New Impeller in Cell Culture A. Microcarrier suspension and distribution in a spinner flask on Day 9; B. Frequency distribution of cell–microcarrier aggregate diameters before and after impeller modification; C. Cell proliferation curves before and after impeller modification; D. Effects on pluripotency genes and differentiation marker genes before and after impeller modification
4.Conclusion and Outlook: Defining the “Golden Rule” of Stem Cell Culture
For mesenchymal stem cells, controlling aggregate size depends less on shear force intensity and more on suspension efficiency. Data show (Figure 6) that at identical shear rates, different impeller designs produce significantly different aggregate sizes. In contrast, Qz—an indicator closely tied to suspension capacity—shows a clear negative correlation with aggregate size: stronger suspension leads to smaller aggregates.
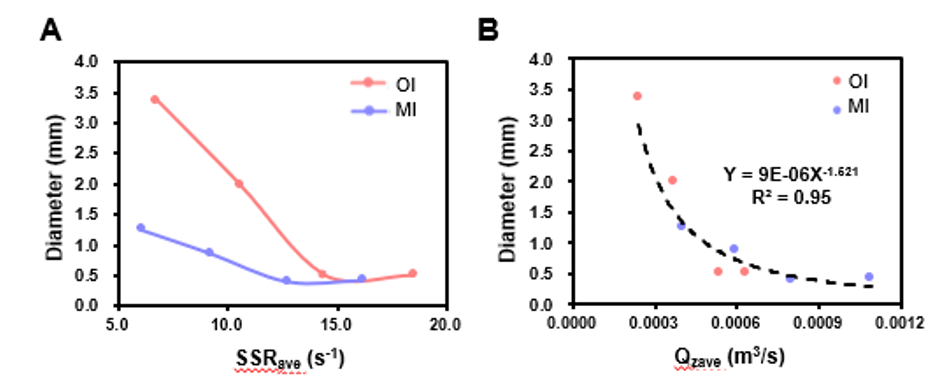
Figure 6. Effects of Shear Force (A) and Qz (B) on Aggregate Size
Guided by this principle, the newly optimized impeller enhances axial suspension through flow-field optimization while maintaining gentle, low-shear conditions, achieving:
· Effective aggregate size control below 1 mm;
· Nearly 40% higher cell density;
· Stable maintenance of stem cell pluripotency.
This impeller upgrade marks a shift from empirical adjustment to science-driven design, providing a practical solution for the large-scale, high-quality in vitro expansion of mesenchymal stem cells. It overcomes key limitations of conventional culture systems and meaningfully advances the translation of regenerative medicine into clinical application. With deep expertise in bioreactor cultivation, Morimatsu will continue supporting mesenchymal stem cell research and development.
Looking ahead, by further integrating computational modeling, intelligent sensing, and process optimization, we aim to build more precise and controllable cell culture systems—helping this “universal repairer” move beyond the lab and bring new hope for tissue repair and regeneration to more patients.
References
[1] Botao Zhang, Qiaohui Lu, Gance Dai, Yi Zhou, Qian Ye, Yan Zhou, Wen-Song Tan. Enhancing Mesenchymal Stem Cells Cultivated on Microcarriers in Spinner Flasks via Impeller Design Optimization for Aggregated Suspensions[J]. Bioresources and Bioprocessing. 2023, 10(1): 89.
[2] Rodríguez-Fuentes D E, Fernández-Garza L E, Samia-Meza J A, et al. Mesenchymal stem cells current clinical applications: a systematic review[J]. Archives of Medical Research, 2021, 52(1): 93-101.
[3] Sart S, Tomasi R F-X, Barizien A, et al. Mapping the structure and biological functions within mesenchymal bodies using microfluidics[J]. Science Advances, 2020, 6(10): eaaw7853.
[4] Liu L, Yu B, Chen J, et al. Different effects of intermittent and continuous fluid shear stresses on osteogenic differentiation of human mesenchymal stem cells[J]. Biomechanics and Modeling in Mechanobiology, 2012, 11(3-4): 391-401.
[5] Higuera G A, van Boxtel A, van Blitterswijk C A, et al. The physics of tissue formation with mesenchymal stem cells[J]. Trends Biotechnol, 2012, 30(11): 583-590.
About Morimatsu LifeSciences
Morimatsu LifeSciences is a key business segment of Morimatsu International Holdings Limited (Stock Code: 2155.HK). It comprises Shanghai Morimatsu Pharmaceutical Equipment Engineering Co., Ltd., Morimatsu (Suzhou) LifeSciences Co., Ltd., Shanghai Morimatsu Biotechnology Co., Ltd., Shanghai Mori-Biounion Technology Co., Ltd., Shanghai Morisora Technology Co., Ltd., Bioengineering AG, Pharmadule Morimatsu AB, and its affiliated companies.
Morimatsu LifeSciences is dedicated in providing core equipment, process systems, and smart modular facility solutions, and services for the pharmaceutical, biopharmaceutical, medical aesthetics, and fast-moving consumer goods (FMCG) sectors including (cosmetics, food, and health supplements), as well as data centers.
Our team comprises highly experienced professionals with deep expertise in process R&D, engineering design, advanced manufacturing, compliance and validation consulting, production execution, and intelligent operations. With broad experience across diverse industries, we fully understand the unique characteristics and process requirements of various products. This enables us to deliver tailored, end-to-end process solutions from the conceptual design stage, precisely aligned with client’s specific needs.
Morimatsu LifeSciences has established a strong global presence, supported by advanced R&D centers, design hubs, and state-of-the-art manufacturing facilities worldwide. Our well-established service network spans Europe,USA,Asia-Pacific, and emerging markets. We have successfully delivered outstanding, customized solutions to clients in over 40 countries and regions, gaining extensive experience in international project execution.
As a multinational enterprise with core strengths in process technology, modular facility construction, and intelligent manufacturing, Morimatsu LifeSciences is dedicated to meeting the evolving equipment and system needs of our key industries. Through continuous innovation and technological advancement, we are steadily expanding our global footprint, driving our international strategy forward, and delivering Morimatsu’s expertise, reliability, and innovation to the global life sciences and related sectors.