
The accuracy of production weighing, data integrity, and compliance control form the cornerstone of high-quality development in the pharmaceutical industry. Against the backdrop of digital transformation and increasingly stringent regulations, traditional manual weighing and recording methods can no longer meet the rigorous demands of modern pharmaceutical companies for quality traceability and compliance management.
Against this backdrop, the weighing management system has emerged as a digital-intelligent, full-process solution for weighing and material tracking, specifically designed for highly compliant production environments. Developed based on a Windows desktop architecture, the system is fully compatible with various weighing and barcode hardware devices. It features core functionalities such as full-process traceability, GMP-compliant design, intelligent weighing guidance, and automated report generation, empowering pharmaceutical enterprises to achieve efficient, compliant, and traceable production management.
1. Pain Point Analysis
Low Operational Efficiency: Predominantly manual operations lead to cumbersome processes and poor coordination; prolonged weighing times and slow material turnover hinder overall production and warehouse flow.
Poor Weighing Accuracy: Manual readings and recordings are prone to errors, compromising data accuracy and affecting cost accounting and production ratios.
Delayed Data Management: Dispersed and isolated data, coupled with non-real-time statistics, leave management decisions lacking effective data support.
High Information Security Risks: Paper records are prone to damage and tampering, while electronic logs lack permission controls and operation trails, making data authenticity difficult to guarantee.
Compliance Traceability Challenges: Absence of a closed-loop traceability system for full-process data, coupled with missing records in critical steps, fails to meet regulatory and audit compliance requirements.
2. Morimatsu Solution
This solution adopts an integrated hardware-software architecture. Through the deep integration of stable and reliable hardware equipment and a mature and secure software system, it builds an efficient, accurate and traceable intelligent weighing management system. Centered on high-precision electronic scales to ensure accurate material weighing, it is equipped with barcode scanners to quickly identify material information, realize identity binding and full-process traceability. The computer acts as the system control and operation center, and the database server centrally stores all service data to ensure data security, stability and traceability.
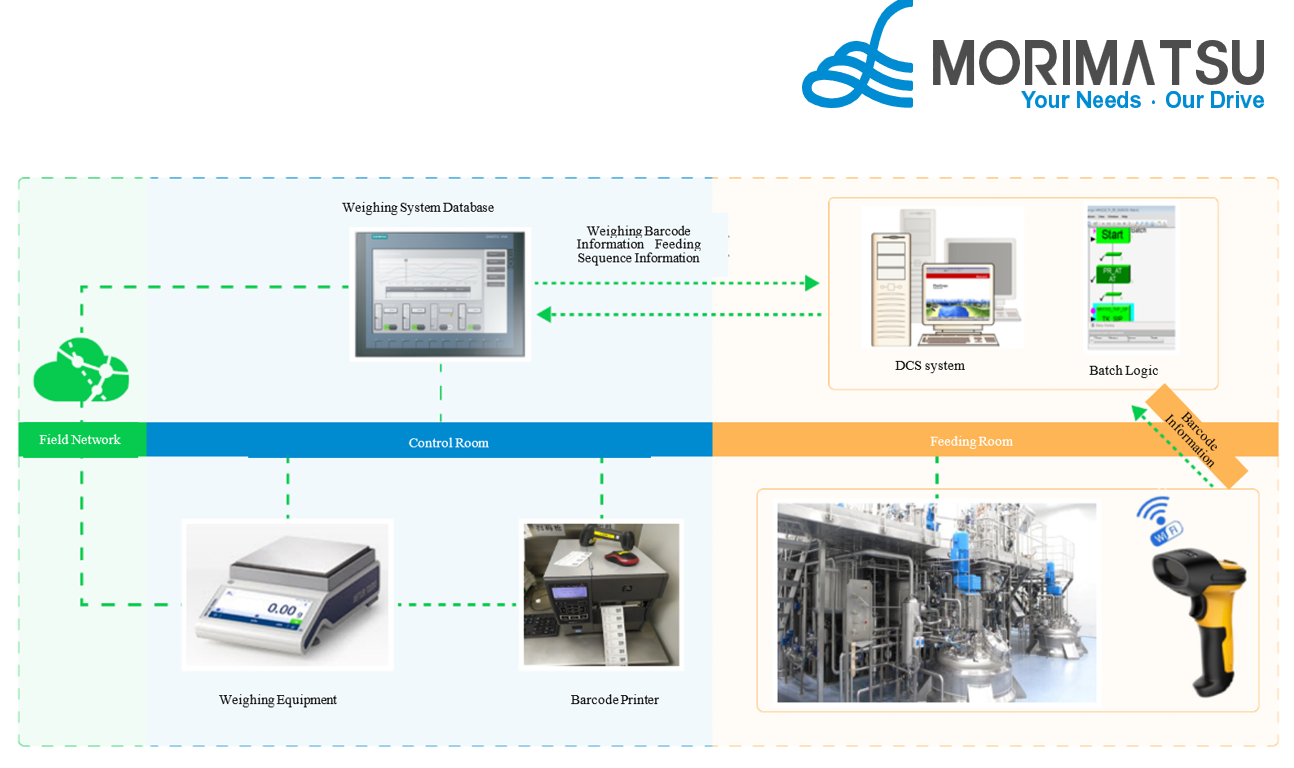
Weighing Management System Diagram
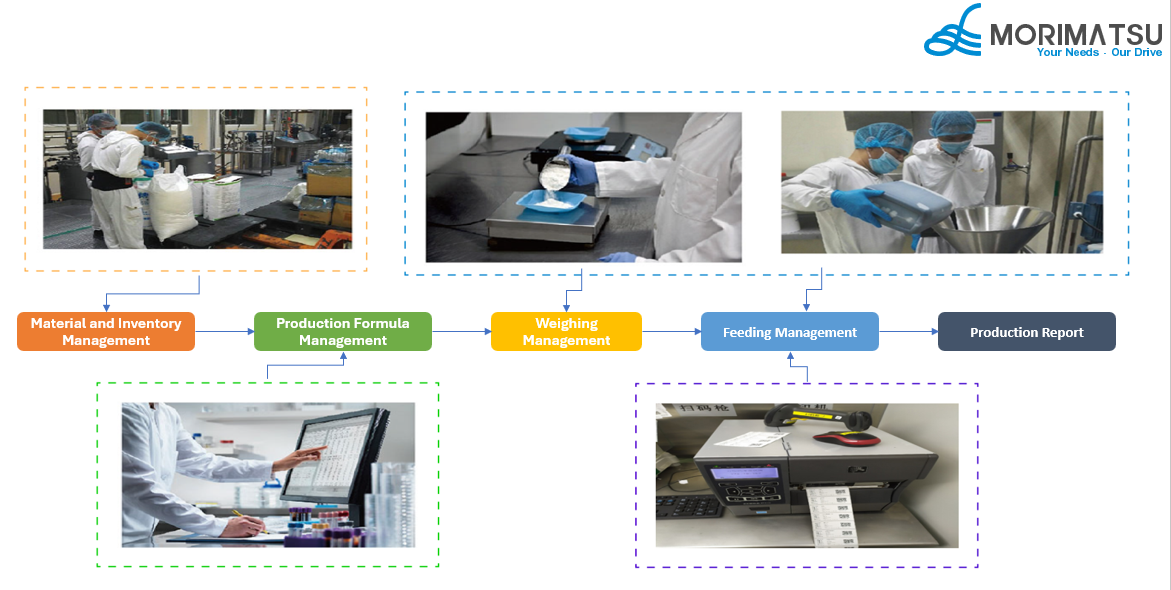
Weighing Management System Flowchart
3. Core Functions of Weighing Management System
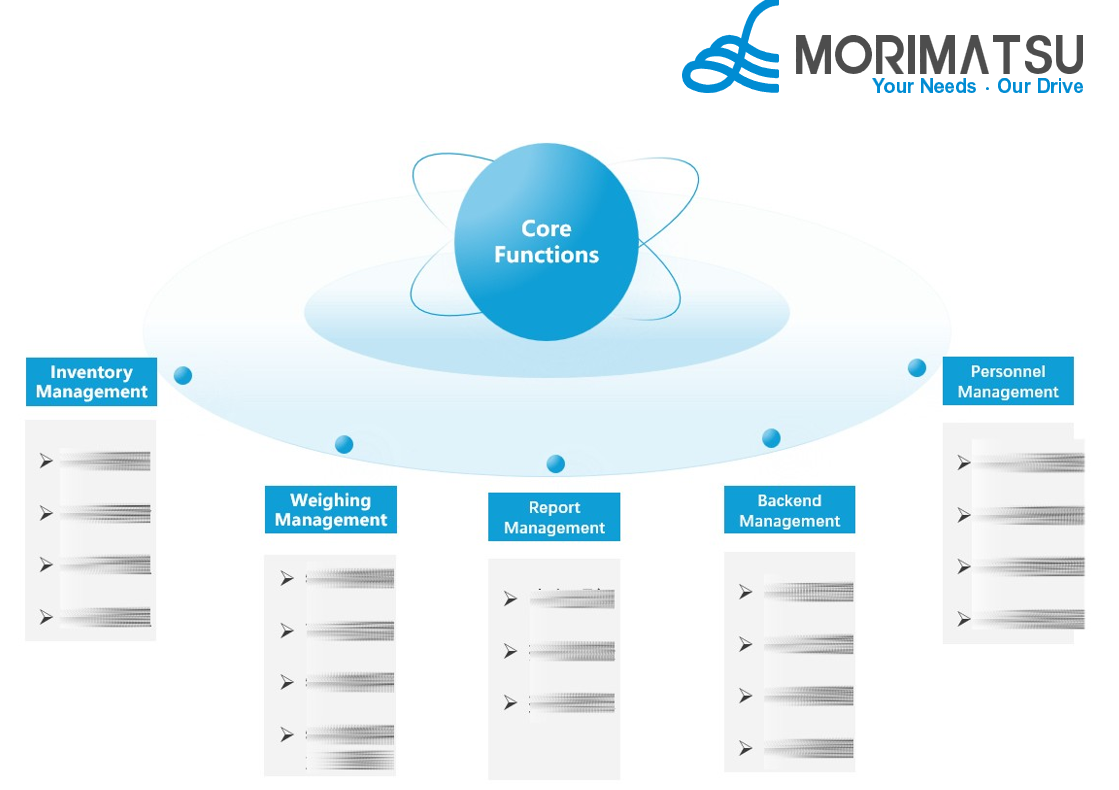
Weighing Management System Architecture Diagram
01 Inventory Management
Material inventory management is a critical aspect of pharmaceutical production and quality control. The system supports barcode printing, real-time inventory query and dynamic update. It allows flexible management of material origin, unit of measurement, packaging specification, upper and lower thresholds, and provides core functions such as material warehousing and material return. It realizes standardized material information, traceable data and controllable inventory, providing solid support for stable production and compliant management.
02 Weighing Management
The weighing management module covers the entire process of weighing operations, pre-and post-site clearance, and material balance calculation. It supports calibration of platform scales and balances as well as calibration history query, and offers various weighing modes including gross weight, net weight, loss-in-weight, automatic potency calculation, and compensation calculation. It enables real-time viewing of material balance data with standardized operations and complete records, fully meeting the compliance control requirements of pharmaceutical production.
03 Report Management
The report management module supports multi-dimensional query, printing and export of material inbound and outbound details and weighing information reports, and provides complete operation logs. It enables data to be queried, traced and exported, offering reliable and authoritative data support for compliance audits and production traceability.
04 Backend Management
The backend management module enables formula access, customized configuration of material calculation formulas, and full-lifecycle management of weighing instruments and equipment (addition, editing, deletion, query), ensuring flexible, accurate and efficient system configuration with strong adaptability.

04 Overall Solution Advantages
Break Through Operational Bottlenecks with Efficient Closed-Loop Processes
Replace manual cumbersome operations with automated weighing, recording, and uploading, significantly reducing weighing time. It bridges the gaps between production and logistics, markedly improving material turnover and overall operational efficiency.
Eliminate Human Errors for Precise and Reliable Weighing
Avoid manual readings and entries with system-automated data collection and rule-based calculations, preventing misrecords, omissions, and misreads at the source. This ensures data accuracy, supporting cost accounting, material control, and precise production ratios.
Centralized and Visualized Data for Informed Decision-Making
Break down data silos by unifying weighing data collection, real-time synchronization, and automated statistics. It creates visualized, multi-dimensional data views and integrates with ERP, MES, etc., providing timely and reliable data support for procurement, sales, and production management.
End-to-End Security and Traceable Operations
Hierarchical access control with audit trails for critical operations. Data is automatically stored, tamper-proof, and loss-resistant, effectively reducing information security risks while ensuring data authenticity, integrity, and security.
Meet Compliance Requirements and Achieve Full-Link Traceability
Establish a closed-loop traceability system covering the entire process from warehousing, weighing, usage to outbound, with key steps fully documented, effortlessly meeting compliance needs for industry regulations, quality traceability, and internal/external audits.
05 Establish a closed-loop traceability system covering the entire process from warehousing, weighing, usage to outbound, with key steps fully documented, effortlessly meeting compliance needs for industry regulations, quality traceability, and internal/external audits.
Morimatsu Weighing System has been implemented in the material management scenario of an enterprise’s plant. With deep integration of multiple systems and full-process digital-intelligent control as its core strengths, it achieves precise, standardized and efficient operation of material management.

Project Case Images
01 Core Advantages
Seamless multi-system integration, full-process digital-intelligent control, dual verification for material input, and highly efficient operations.
02 System Configuration
Each weighing room is uniformly equipped with floor scales, balances, label printers, and touchscreen interfaces for localized convenient operations.
03 Core Functions
Multi-system Integration: Deep integration with SAP enables bidirectional data read/write and real-time synchronization, eliminating information silos;
Intelligent Data Binding: Post-weighing, materials are precisely labeled via on-demand printing for accurate information binding;
Precision Dosing Control: Supports formula-compliant material dispensing with barcode scanner verification to prevent dosing errors;
End-to-end Management: One-stop solution for formula management, material dispensing, inventory balancing, and returns, forming a closed-loop material control system.
04 Application Value
Consolidate the full-link management and control system for plant materials, comprehensively improve material management efficiency and control accuracy, and lay a solid foundation for the enterprise's digital-intelligent transformation.
Morimatsu's Digital Weighing Management System delivers an all-in-one solution for pharmaceutical weighing challenges, enabling efficient, compliant, and traceable material management through full-process digitalization.
About Morimatsu LifeSciences
Morimatsu LifeSciences is one of the key business segments of Morimatsu International Holdings Company Limited (Morimatsu International, Stock Code: 2155.HK). It primarily consists of Shanghai Morimatsu Pharmaceutical Equipment Engineering Co., Ltd., Morimatsu (Suzhou) LifeSciences Company Limited, Shanghai Morimatsu Biotechnology Co., Ltd., Shanghai Mori-Biounion Technology Co., Ltd., Shanghai MoriSora Technology Co., Ltd., Bioengineering AG and Pharmadule Morimatsu AB, along with their subsidiaries.
We specialize in providing core equipment, process systems, and comprehensive digital-intelligent plant solutions along with corresponding services for industries including pharmaceuticals, biopharmaceuticals, medical aesthetics, fast-moving consumer goods (FMCG, including cosmetics, food, health products, etc.), and data centers. The company boasts an elite team composed of seasoned experts across multiple fields, including process R&D, engineering design, high-end manufacturing, validation consulting, production execution, and digital-intelligent operations. The team members possess extensive experience in industries such as pharmaceuticals, biopharmaceuticals, medical aesthetics, FMCG, and data centers. They are well-versed in the characteristics and process flows of diverse products, enabling them to provide customized process solutions tailored to client needs from the conceptual design stage onward.
Morimatsu LifeSciences has established a global strategic presence, with advanced R&D, design centers, and manufacturing bases across different countries and regions, complemented by a comprehensive service network covering Europe, America, Asia-Pacific, and emerging markets. The company's international professional team has successfully delivered customized solutions to over 40 countries and regions, accumulating rich experience in global project execution.
As a multinational enterprise mastering core process technologies, modular construction, and digital intelligent plant expertise, Morimatsu LifeSciences is committed to meeting the production equipment demands of global industries, including pharmaceuticals, biopharmaceuticals, medical aesthetics, FMCG, and data centers. Through continuous innovation and optimization, the company supports domestic enterprises in accelerating the strategic localization of core and high-end equipment, driving industry advancement. Simultaneously, the company actively expands overseas markets, deepening its "globalization" business development strategy to contribute Morimatsu's expertise to the global life sciences and related industries.